|
|||||||||||||||||||||||
|
ü@ü@We recently had an opportunity to examine faceted stones in orange and blue colours as shown on the photo 1. They appeared at a glance just as fire opal from Mexico and blue opal from Peru, however, our inspection revealed that they were artificial products. From our succeeding study, they were proved to be synthetic opals manufactured by RMC Gems Thai Co Ltd., and they have been on sale since 2007 under the commercial names ügMexiFireüh and ügPeruBluüh, named after important locality of natural opals (Jewellery News Asia, November 2007). The gemological features of these synthetic opals are introduced here.
ü@ü@The stones tested this time were one piece of faceted fire opal and two pieces of faceted blue opals. The fire opal displayed orange color, with high transparency and vitreous luster, but no play of color was shown. Its appearance closely resembled a natural fire opal in good quality without fractures from Mexico. The blue opals, displaying extremely highly saturated blue color such as Para?ba tourmaline that is often expressed as ügelectric blueüh, had higher transparency than natural opal from Peru that were generally translucent to opaque and characteristically contained no fractures or cracks. ü@ü@Gemological physical properties of these stones were tested and RI and SG (by hydrostatic weighing method) measured 1.35 and 1.57 respectively in fire opal, while those measured 1.39 and 1.75 similarly in blue opal. Any of these properties was lower than those of natural fire opal from Mexico or natural blue opal from Peru, and it can be an important feature in distinguishing them from natural opal. Anomalous extinction reaction was recognized in all the stones between crossed polarizing filters. Under magnification observation, densely spread minute inclusions that were forming clouds and gas bubbles were easily recognized throughout the stones (photo 2). Fine wavy growth structure, which is not seen in natural opal, was also recognized (photo 3). These synthetic opals we have tested this time did not show üglizard skinüh that is characteristically seen in synthetic opals manufactured by Kyocera or Gilson and thus they are assumedly produced by a new synthesis. The synthetic fire opal displayed faint blueish white color under long wave UV light and faint chalky yellow-green color under sort wave UV light in some area, while the synthetic blue opals were inert both under long and short wave UV lights. All the stones showed no change through a color filter. With a hand-held spectroscope, an absorption starting at 500 nm towards shorter wavelength side was observed in the synthetic fire opal, whereas no distinct absorption other than weak absorption in red area was observed in the synthetic blue opals.
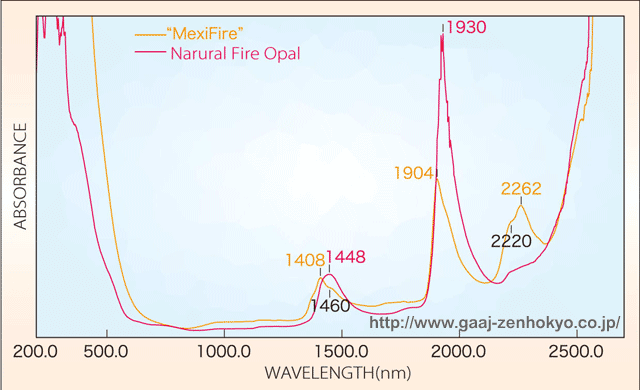
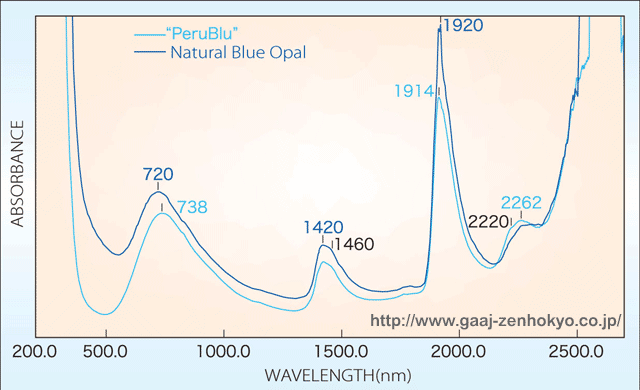
ü@ü@ spectrum measurement by a spectrophotometer indicated no distinct difference between the synthetic opals tested this time and natural opals in UV-visible region. On the other hand, in near-infrared region, the spectra of these synthetic opals showed absorptions associated with H2O or OH that usually cannot be seen in general synthetic opals, and these spectra closely resembles those of natural opals. However, natural fire opals show strong absorption peaks centered on about 1450 and 1930 nm, while these synthetic opals showed the peaks shifted on to about 1410 and 1900 nm, with another strong absorption at about 2260 nm (figure 1 and 2).
ü@ü@On an infrared spectrophotometry with FTIR, transmitted and reflected spectra of these synthetic opals appeared quite similar to those of common natural opals. A further inspection on absorption peaks in the transmitted spectra revealed that natural fire opal showed weak absorption peak at about 4500 cm-1, while the synthetic fire opal showed the strongest peak at about 4420 cm-1 and a weaker absorption at about 4500 cm-1. ü@ü@In a compositional analysis by a fluorescent X-ray analysis device, other than the main component Si, trace of Fe and Cu were detected from the synthetic fire opal and the synthetic blue opals respectively, but both materials did not indicate any other characteristic elements. The further analysis on trace elements in both materials by LA-ICP-MS detected elements such as B, Na, Mg, Al, K, Ca, Sc, Ti, Cr, Mn, Ni, Zn, Sr, Rh, Sn and Pb, other than Fe and Cu, the colouring elements of these opals. Natural fire opals and natural blue opals contain trace elements such as Be, V, Ga, Sr and U, which were not detected in this type of synthetic opals, and our study also showed differences of the contents of the elements such as Mg, Al, K, Ca and Zn between natural opals and this type of synthetic opals. ü”The synthetic opals tested this time were courtesy of MIYUKI CO.,LTD., and we express here our sincere gratitude. |
|||||||||||||||||||||||