|
||||||||||||||||||||||||||||||||||
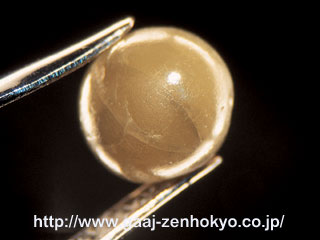
| Along with the diversifying demand for gem materials, our opportunity to meet unusual gemstones during practical gem identification work has been increasing. Some of the materials which draw attention in the industry are introduced here. This report is based on a lecture presented at the general lecture in annual conference of the Gemmological Society of Japan 2005, which was partly added and amended. ♦ Black Diamond Black diamond has become very popular for jewellery use since the end of 1990s and many jewellery pieces set with a number of small black diamonds are commonly seen in our identification work nowadays. This natural black colour does not come from the body of a stone, but in most cases, black inclusion contained in pale brown to pale yellow body colour makes a stone appear black. The black inclusion consists of granular or needle-like graphite or sulphide minerals. Growing demand for black diamond required securing of sufficient material, and it made substitute with artificial irradiation come appear. This material is obtained mainly by irradiating neutron in an atomic reactor, and although it appears black, it is actually deep blue to green. Thus the material tends to appear blue to green against strong transmitted light such as fibre optic light. A new type of Ågblack" diamond started to appear on the market several years ago. This is generally called HT (high temperature)-treated diamond. The stone is originally natural diamonds of very poor quality single crystal or white to grey polycrystalline crystal, which is turned to black by being heated in high temperature (HT) under reducing atmosphere (i.e., lacking in oxygen condition) to graphitise partly (photo-1).
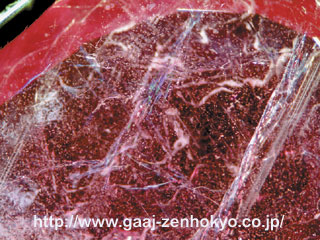
To distinguish such HT-treated black diamond, careful magnification test using a gemmological microscope is essential. Needle-like black inclusions are identifying feature of natural black diamond, and black inclusions in irregular form are scattered inside a natural black diamond. Contrary to this, black inclusions in HT-treated diamond tend to be concentrated near surface or in surface-reaching fracture (photo 2). In addition, photoluminescence (PL) analysis that is effective to detect HPHT treatment can be applied to complement those visual inspections above.
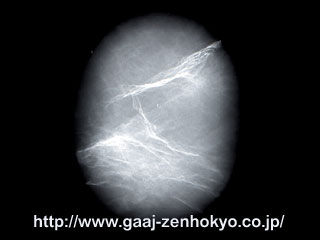
♦Ruby with lead-glass filling Treating diamonds by impregnating its cleavages or fractures with highly refractive glass to improve the clarity (transparency) has been commonly known. This type of treatment appeared on the market around 1987 and has been called Ågglass filling" in overseas. It also is called ÅgKoss" treatment or ÅgYefuda" treatment, named after the treaters. Diamonds treated in this manner typically show unique rainbow-coloured light in their cleavages and fractures, and it is called flash effect. This flash effect has been seen in some faceted ruby since last year (photo 3). Residues from heating process may often be seen in fractures and cavities in ruby, but they have far more low refractive indices than ruby. Thus, it never produces flash effect. Flash effect seen in ruby indicates that the stone is impregnated with substances whose refractive indices overlap with that of ruby. In the radiograph of X-ray test, many white streaks in high contrast are seen. They correspond to distributed fracture developed in ruby and indicate existence of elements which have larger atomic mass than the host ruby (Al2O3) (photo 4). Further test on the surface reaching fractures by X-ray fluorescence analysis detected Pb (lead) other than the main chemical element of ruby, Al, and common trace elements. This type of lead-glass filling treatment is mainly seen in large faceted ruby, however, it is also seen in cabochon star stone and small pieces of beads in a necklace. Unlike in colourless transparent diamond, flash effect in fracture of ruby may be masked by the body colour and be difficult to detect. When determination cannot be made by such visual observation, confirmation with X-ray radiograph test or X-ray fluorescence analysis is required. *2 In addition, Be-diffusion heat treatment and lead-glass infilling treatment are performed on a same stone most recently, so that more caution should be taken in Identification of ruby
♦Violet Coloured Gem Minerals Violet gemstones are designated as the Year Colour Stone of 2005 by JJA (Japan Jewellery Association). We introduce here some of unusual violet gemstones (photo 5).
Violet Chalcedony This was newly discovered in the east part of Sumatra in Indonesia recently. The stone shows characteristic deep violet colour that reminds of amethyst and violet sapphire. It has high transparency and saturation comparing to amethystine chalcedony that is a traditional variety of chalcedony, and it shows colour patches that is commonly seen in agate under magnification test. The cause of the violet colour is assumed a colour centre related to iron, resembling amethyst, by observation of transition of colour hue in X-ray fluorescence analysis and in colour-fading test. Stichtite Stichtite, one of rare gemstones, was brought in our laboratory for identification. This is a metamorphic mineral of serpentinite containing Mg and Cr, and it should be handled with care because its hardness is low as 1.5Å`2 on the Mohs' scale. The stone has the appearance of sugilite but their chemical composition and crystal structure are different so that they can be distinguished from each other by X-ray fluorescence analysis or Raman spectroscopy. Violet Turquoise Violet turquoise was sold at the Tucson Gem & Mineral Show 2005. According to the dealer, it was a heated and pressed turquoise from Arizona, and during the treatment process the material changed into violet colour, which is derived from manganese. However, compositional analysis by X-ray fluorescence test revealed that any element relating to colouring was not detected. Cermikite This gemstone was also sold at the Tucson Gem & Mineral Show 2005. The clear crystal resembles a large piece of fluorite, but actually this is allegedly an artificial crystal of coloured chromium alum or colourless alum produced in China. A sample of similar type that GAAJ purchased several years ago appeared as a cluster of amethyst at the time, however, now it has faded to colourless. Consequently, this Cermikite may fade its colour across the ages. *3 |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||